

|
Author(s)
Steven Percival
Philip Bowler
|
Contents
|
|
Published:
July 2004
Last updated: June 2004 Revision: 1.0 |
Keywords: biofilms; bacterial communities; critical colonisation; wound infection.
Communities of micro-organisms are likely to exist within their own microenvironment (biofilm) in wounds
Microbial interactions and biofilms may have a significant effect on wound healing and infection
Further research is needed to facilitate deeper understanding of the relationship between biofilm communities and wound pathophysiology in order to promote wound healing and infection control.
For survival and reproductive success, species of bacteria often rely on close relationships with other species. A collection of bacteria occupying the same physical habitat is called a 'community', and one example of a community of micro-organisms is a 'biofilm'. Biofilms have been implicated in numerous chronic infections including cystic fibrosis, otitis media and prostatitis. Through interactions within a biofilm, the resident population of bacteria is likely to benefit from increased metabolic efficiency, substrate accessibility, enhanced resistance to environmental stress and inhibitors, and an increased ability to cause infection and disease. Dermal wounds often provide an ideal environment for bacteria to exist as a community, which may have a significant effect on wound healing.
The conditions under which species of micro-organisms can survive in nature are determined by physiological and ultimately genetic competence. Consequently species of bacteria often rely on close relationships with other species for survival and reproductive success. A collection of bacteria occupying the same physical habitat is called a 'community', and one example of a community of micro-organisms is a 'biofilm' [1]. A biofilm forms when bacteria attach to a surface and subsequently encase themselves in an exopolymeric material [1]. Such bacteria are morphologically and physiologically different from free-living planktonic bacteria and have been implicated in numerous chronic infections ranging from cystic fibrosis to prostatitis [1], [2]. The existence of biofilms in an acute partial-thickness wound [3] and in chronic human wounds [4] has been documented.
The relationship between a micro-organism and a specific disease is described by Koch [5], [6] who states that a micro-organism must be present in every case of the disease, isolated and grown in pure culture, and then shown to cause the same specific disease when inoculated into a healthy host with the same micro-organism isolated again from the diseased host. However, this situation does not apply where a community of organisms are collectively associated with an infection, particularly when specific organisms are not consistently causative.
Chronic wounds are invariably polymicrobial, yet most research to date has focused on the role of specific potential pathogens in wounds (eg Pseudomonas aeruginosa) rather than the effect of interactions between different species. Only recently have microbiologists begun to address and recognise the significance and importance of an interacting community and the relationship to the disease process. It is the intention of this review to address the potential role that microbial communities and associated biofilms may play in wound healing.
The composition (chemical, biological and physical) of a surface will determine the initial microbiology that develops at that surface. In the case of a recently formed wound, different micro-organisms from endogenous and exogenous sources will contaminate the wound surface. The properties of the wound surface will predetermine which micro-organisms will attach, grow and remain components of an early biofilm. The initial colonising bacteria on any surface are referred to as the 'pioneering' species. Colonising bacteria will modify the habitat and create a 'microenvironment' that encourages the attachment and growth of secondary colonising micro-organisms. If unchallenged, and with favourable conditions, a complex community of micro-organisms is likely to develop. Although such a continuum of events has been documented in the oral cavity, microbial development and succession in wounds have not been investigated. It has been proposed that as a microbial biofilm develops the community will ultimately form a more stable 'climax community' [7]. As the community is able to adapt appropriately to outside perturbations the term 'microbial homeostasis' has been suggested to reflect stability within a 'climax community' [7].
Within a wound environment, and particularly in the presence of devitalised sloughy tissue, obligate anaerobes are one of the dominant groups of micro-organisms, despite the frequent exposure of the wound to air [8]. Anaerobes are able to cope with the toxic effects of oxygen by interacting with bacteria that are able to grow in air. As the aerobic bacteria grow they consume oxygen and create a more favourable environment for anaerobic bacteria. This has been demonstrated in laboratory studies involving communities of oral bacteria [9], [10], [11].
Many bacteria have a relatively narrow pH range for growth. When present as a community within a wound biofilm, these bacteria are able to survive within a pH range that would be inhibitory to cells growing in pure culture [9]. Therefore microbial communities are able to overcome the constraints imposed by the external macro-environment by creating, through their metabolism, a mosaic of microenvironments that enable the survival and growth of the component species. Similarly the sequential breakdown of various nutrients within a polymicrobial community leads to the formation of a simple food chain whereby the product of one organism will become the substrate of another organism. This process will ultimately lead to the development of microbial homeostasis within the community as the component species would rely on one another to provide substrates for growth and development.
Most infections are caused by a combination of micro-organisms (polymicrobial or mixed infection) [12]. In a wound environment, individual organisms may be unable to satisfy the requirements to cause disease and subsequently combine forces to do this. Examples of this type of situation have been documented for diseases of the gum and peridontium [13] and leg ulcer infections [8].
In many animal models numerous combinations of anaerobic and aerobic bacteria have been shown to produce levels of sepsis or disease that could not be induced by individual species [13]. Such synergy has been demonstrated in wound pathogens such as: Prevotella melaninogenicus, Porphyromonas asaccharolytica and Peptostreptococcus micros [14]; Porphyromonas asaccharolytica and Klebsiella pneumoniae [8], [15]; Escherichia coli and Bacteriodes fragilis [8], [16] and Staphylococcus aureus [8].
Mechanisms that constitute pathogenic synergy include communal protection from phagocytosis and intracellular killing, production of essential growth factors, modification of the local environment (eg redox potential) and the protection of sensitive species by the inactivation of inhibitors. This protection has been named 'indirect pathogenicity' [17]. In certain situations some pathogens are found to be antibiotic sensitive but are rendered 'resistant' by other members of the mixed infection. Treatment in these cases would be targeted at all of the component micro-organisms in a mixed infection [17], [18].
Opportunistic pathogens are members of the resident microflora that become pathogenic by gaining access to sites that are not normally accessible to them, or by causing changes in the local environmental conditions that favour their prevalence within a community. In addition to synergy, organisms within a wound may interact via quorum sensing. Quorum sensing involves communication strategies within a mixed community of organisms that ultimately enable them to coordinate their activities, and enhance their pathogenicity and ability to cause disease.
Wound healing and infection is influenced by the relationship between the ability of bacteria to create a stable, prosperous community within a wound environment and the ability of the host to control the bacterial community. Since bacteria are rapidly able to form their own protective microenvironment (biofilm) following their attachment to a surface, the ability of the host to control these organisms is likely to decrease as the biofilm community matures. Within a stable, climax biofilm community, interactions between aerobic and anerobic bacteria are likely to increase their net pathogenic effect, enhancing their potential to cause infection and delay healing.
As well as the capability to have a direct effect on wound healing through the production of destructive enzymes and toxins, mixed communities of organisms may also indirectly affect healing by promoting a chronic inflammatory state (see Figure 1). It is apparent that prolonged exposure to bacteria within a chronic wound leads to a prolonged inflammatory response, resulting in the release of free radicals and numerous lytic enzymes which could have a detrimental effect on cellular processes involved in wound healing. Proteinases released from a number of bacteria, particularly Pseudomonas aeruginosa, are known to affect growth factors and many other tissue proteins that are necessary for the wound healing process [19], [20]. The increased production of exudates that often accompanies increased microbial load has been associated with the degradation of growth factors and matrix metalloproteinases (MMPs) which subsequently affect cell proliferation and wound healing [21].
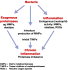
In summary, biofilm communities and associated bacterial interactions have been poorly researched in relation to wound healing, but it is likely that their effect on the wound healing process, through both direct and indirect mechanisms, is significant. Further research in this area is important to understand the relationships between biofilm communities, wound pathophysiology, infection and healing.
1. Costerton JW, Stewart PS, Greenberg EP. Bacterial biofilms: a common cause of persistent infections. Science 1999; 284(5418): 1318-22.
2. Costerton JW, Lewandowski Z, Caldwell DE, Korber DR, Lappin-Scott HM. Microbial biofilms. Annu Rev Microbiol 1995; 49: 711-45.
3. Serralta VW, Harrison-Belestra C, Cazzaniga AL, Davis SC, Mertz PM. Lifestyles of bacteria in wounds: presence of biofilms? Wounds 2001; 13(1): 29-34.
4. Bello YM, Falabella AF, Cazzaniga AL, Harrison-Balestra C, Mertz PM. Are biofilms present in human chronic wounds? Presented at the Symposium on Advanced Wound Care and Medical Research Forum on Wound Repair, Las Vegas, NV. April, 2001.
5. Koch R. Methods for the study of pathogenic organisms. Mittheilungen aus dem Kaiserlichen Gesundheitsamte 1881; 1: 7-48.
6. Koch R. The etiology of tuberculosis. Mittheilungen aus dem Kaiserlichen Gesundheitsamte 1884; 2: 1-88.
7. Alexandra M. Microbial Ecology. New York: Wiley, 1971.
8. Bowler PG, Duerden BI, Armstrong DG. Wound microbiology and associated approaches to wound management. Clin Microbiol Rev 2001; 14(2): 244-269.
9. Bradshaw DJ, Marsh PD, Allison C, Schilling KM. Effect of oxygen, inoculum composition and flow rate on development of mixed culutre oral biofilms. Microbiology 1996; 142: 623-629.
10. Bradshaw DJ, Marsh PD, Watson GK, Allison C. Oral anaerobes cannot survive oxygen stress without interacting with aerobic/facultative species as a microbial community. Lett Appl Microbiol 1997; 25: 385-387.
11. Bradshaw DJ, Marsh PD, Watson GK, Allison C. Role of Fusobacterium nucleatum and coaggregation in anaerobe survival in planktonic and biofilm oral microbial communities during aeration. Infect Immun 1998; 66(10): 4729-32.
12. Jenkinson HF, Dymock D. The microbiology of periodontal disease. Dent Update 1999; 26(5): 191-97.
13. Brook I. Role of encapsulated anaerobic bacteria in synergistic infections. Crit Rev Microbiol 1987; 14(3): 171-93.
14. Sundqvist GK, Eckerbom MI, Larsson AP, Sjögren UT. Capacity of anaerobic bacteria from necrotic dental pulps to induce purulent infections. Infect Immun 1979; 25(2): 685-93.
15. Mayrand D, McBride BC. Exological relationships of bacteria involved in a simple, mixed anaerobic infection. Infect Immun 1980; 27(1): 44-50.
16. Rotstein OD, Kao J. The spectrum of Escherichia coli-Bacteroides fragilis pathogenic synergy in an intraabdominal infection model. Can J Microbiol 1988; 34(3): 352-57.
17. Brook I. Direct and indirect pathogenicity of beta-lactamase-producing bacteria in mixed infections in children. Crit Rev Microbiol 1989; 16(3): 161-80.
18. Brook I. Microbiology and management of sinusitis. J Otolaryngol 1996; 25(4): 249-56.
19. Steed DL, Donohoe D, Webster MW, Lindsley L. Effect of extensive debridement and treatment on the healing of diabetic foot ulcers. Diabetic Study Group. J Am Coll Surg 1996; 183(1): 61-64.
20. Travis J, Potempa J, Maeda H. Are bacterial proteinases pathogenic factors? Trends Microbiol 1995; 3(10): 405-07.
21. Falanga V, Grinnell F, Gilchrest B, Maddox YT, Moshell A. Workshop on the pathogenesis of chronic wounds. J Invest Dermatol 1994; 102(1): 125-27.