

|
Author(s)
Douglas Bowley
Kenneth Boffard
|
Contents
|
|
Published:
Oct 2002
Last updated: Oct 2002 Revision: 1.0 |
Keywords: motor vehicle accidents; injury patterns; blunt trauma; management.
There are characteristic injury patterns associated with injuries from road traffic accidents. These injuries are usually multisystem.
Optimal care of severely injured patients requires a coordinated approach, from the point of injury through to rehabilitation.
It is important that practitioners adopt a logical sequence for the initial assessment and management of trauma patients to reduce morbidity and mortality.
Motor vehicle accidents (MVAs) are a major cause of both internal and external wounds, many of which cannot be treated with simple dressings - if at all. In this article the authors discuss the importance of recognising the typical patterns of injury associated with MVAs. This, coupled with a logical sequence for the initial assessment and management of trauma patients, has been shown to contribute to improved outcomes. However, for there to be any significant reduction in the worldwide burden of motor vehicle-related trauma, injury prevention strategies are needed; prevention being better than cure.
In 1990, about 5 million people died worldwide as a result of injury [1]. It is estimated that by the year 2020, 8.4 million people will die every year from injury, and injuries from road traffic accidents will be the third most common cause of disability worldwide and the second most common cause in the developing world [2].

In 2001, over 18,000 patients attended the Johannesburg Hospital Trauma Unit and approximately 140 priority-one casualties were treated per month. In that year there were 1715 resuscitations for trauma, 688 for blunt trauma, of which the majority were associated with road traffic injury. There are characteristic injury patterns, with multisystem injury being the rule rather than the exception.
Optimal care of severely injured patients requires a coordinated approach from the point of injury, through a hospital facility organised to cope with the demands of looking after multisystem problems, to a rehabilitation structure that can return the patient to his or her maximum potential level of function within society. Although sophisticated prehospital and trauma centre systems have been shown to reduce the number of preventable deaths after trauma [3], maximum impact in reducing the burden of trauma must come from injury prevention strategies.
Motor vehicle-related injuries can be arbitrarily divided into:
Collision between the patient and the external environment (the vehicle, or a stationary object if the occupant has been ejected)
Acceleration or deceleration forces acting on the patient's internal organs
In a frontal collision, the unrestrained occupant of a vehicle continues to move forward as the vehicle comes to an abrupt stop. This forward motion is arrested as the patient connects with the, by now, stationary vehicle chassis. The initial impact point is often the lower extremities, resulting in fracture/dislocation of the ankles, knee or hip dislocations and femoral fractures. As the body continues moving, the head, cervical spine and torso impact on the windscreen and steering column.
In a lateral impact, the victim is accelerated away from the side of the vehicle. Compressive pelvic injuries, pulmonary contusion, intra-abdominal solid organ injury and diaphragmatic rupture are common. Rear impacts also accelerate the victim; if head restraints are incorrectly fitted, the inertia of the head makes the cervical spine vulnerable to injury. Ejection from a vehicle is associated with a significantly greater incidence of severe or critical injury[4].
Such injuries occur when differential movement occurs between adjacent structures; for example the distal aorta is anchored to the thoracic spine and decelerates much more quickly than the relatively mobile aortic arch. Shear forces are generated in the aorta by the continuing forward motion of the arch in respect to the distal thoracic aorta. Similar situations occur at the renal pedicles, the junction of the cervical with the thoracic spine and also between the white and grey matter within the brain. The use of a three-point lap-shoulder seat belt is thought to reduce the risk of death or serious injury for front-seat occupants by approximately 45%[5].
In purely frontal MVAs, air bags provide a reduced risk of fatality of approximately 30%. In all crashes the reduction in risk of death has been estimated at 11% [6].
Motorcyclists experience a death rate 35 times greater than the occupants of cars [5]; most of the injuries are to the head and the use of helmets reduces the risk of fatal head injury about one third [7], and the risk of facial injury by two thirds[8]. Fractures of the lower extremities are also common in motorcyclists, occurring in approximately 40% of motorcyclists hospitalised for non-fatal injuries[9].
Injuries due to bicycles account for 800 deaths and approximately 500,000 injuries treated at hospitals per year in the USA[5]. Children aged 5-14 years have the highest rates of injury and head injury accounts for 75% of the deaths associated with bicycling [4]. Helmets have been shown to reduce the risk of brain injury by 88%[10].
Injuries to pedestrians occur disproportionately among the young (particularly school-aged children), the elderly and the intoxicated [4]. Three impact phases are described during pedestrian injury:
Vehicular bumper impact: in the upright adult, initial impact is usually on the lower limbs
Vehicular windscreen impact: torso and head injuries occur as the pedestrian impacts the body of the vehicle
Ground impact: head, spinal and other collision injuries occur as the displaced pedestrian hits the ground.
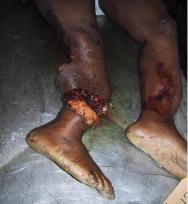
Alcohol and other forms of substance abuse are major associated factors in motor vehicle-related trauma. In South Africa, 7% of drivers with illegal blood alcohol levels account for nearly 30% of non-fatal and 47% of fatal driver deaths [11], while injury to drunken pedestrians account for 72% of adult traffic deaths [11].
In trauma units around the world there is an increasing realisation that trauma can have marked and sustained psychological effects. Up to 25% of severely injured patients experience significant early psychological reactions after trauma[12]. In some patients, these reactions can be long lasting and have profoundly adverse effects on quality of life. Those at high risk can be identified, and effective treatments are available [13]. All practitioners working with injured patients should be sensitive to the psychological effects of major injury and be prepared to offer support and know when to refer for specialist treatment.
In the western world, the most common cause of death after trauma is severe brain injury. The incidence of death from head injury is approximately 7 per 100,000 [14], and the severely brain-injured also have the highest mean length of stay and mean hospital costs [15]. In the European Brain Injury Consortium (EBIC) study, 52% of head injuries were related to MVAs [16]. Head injury is a major cause of morbidity in survivors; disability may occur whatever the initial severity of the head injury and surviving patients with brain injury are more impaired than patients with injuries to other parts of the body. In a prospective study of nearly 3000 head injuries from Scotland, patients were stratified according to the Glasgow Coma Score (GCS) on arrival at hospital: mild injury (GCS 13-15), moderate injury (GCS 9-12) and severe injury (GCS 3-8). Of the initial cohort, 2668 had mild injuries, 133 moderate injuries, and 102 had severe injuries. At follow-up after one year, 1397 were still disabled. Of these, 1260 (90%) had been initially assessed as mild injuries[17].
There are two major categories of brain injury: focal injuries and diffuse injuries.
Focal brain injuries are usually caused by direct blows to the head, and comprise contusions, brain lacerations, and haemorrhage leading to the formation of haematoma in the extradural, subarachnoid, subdural, or intracerebral compartments within the head. Diffuse brain injuries, are usually caused by a sudden movement of the head, and cause diffuse axonal injury.
The availability of computed tomography (CT) scanning has been shown to reduce mortality in patients with acute extradural haematoma[18], as the time taken to diagnose and evacuate an intracerebral haematoma is critical in determining outcome. However, the majority of patients with brain injury do not have a lesion suitable for neurosurgical intervention. Recent guidelines have been produced in an attempt to improve outcome after severe traumatic brain injury [14]. An understanding of the concept of secondary brain injury, caused by hypotension and hypoxia is fundamental and the treatment of a head-injured patient should emphasise early control of the airway (while immobilising the cervical spine), ensuring adequate ventilation and oxygenation, correcting hypovolaemia and prompt imaging by CT.
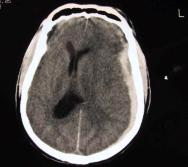
The United States Major Trauma Outcome Study estimated the incidence of acute spinal-cord injury to be 2.6% of blunt trauma patients [19]. The average lifetime costs of treating an individual with traumatic spinal-cord injury is estimated to be between US$500 000 and US$2 million. The total direct costs of caring for the spinally-injured each year exceed US$7 billion in America [20].
Initial mechanical trauma includes traction and compression forces. Events initiated by the primary injury cause extension of the injury to the spinal cord; damaged tissue releases toxic mediators, which damage intact cells; in particular, the chemical, glutamate, overexcites neighbouring neurons and initiates free-radical-mediated cell damage [20]. This process is potentiated by systemic hypotension and hypoxia.
Treatment for spinal injury should focus on prevention of secondary damage; however, the National Acute Spinal Cord Injury Study (NASCIS) phase III trial showed that high dose steroids given within 8 hours of injury reduced disability[21]. The place for early surgery is still unclear, unless the canal is severely compromised. Rehabilitation after spinal injury requires a multidisciplinary approach to reduce morbidity from the psychological and physical problems attendant with spinal cord injury[20]. This type of rehabilitation demands huge resources, which is often beyond the reach of developing countries.
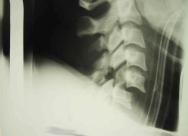
In one series from the USA, the incidence of blunt carotid injuries was 1 in every 150 MVAs [22]. Injury to the carotid and vertebral arteries after blunt trauma can lead to severe neurological complications in survivors and a mortality rate of 31% [23]. A high index of suspicion for these injuries should be maintained as early treatment with systemic heparinisation leads to improved neurological outcome[22]. Also, screening of high risk patients can identify blunt cervical vascular injury in asymptomatic patients and heparinisation is thought to prevent thromboembolisation arising from the intimal injury [23]. Blunt cervical vascular injury should be suspected in patients with:
a major mechanism of injury and unexplained paresis
unequal pupils
cerebrovascular accident or transient ischaemic attack
in the presence of a base of skull fracture, major facial or cervical spinal injury [23].
Diagnosis is by cervical angiography, although non-invasive techniques, such as duplex Doppler or CT angiography may also be used.
In modern day civilian trauma centres, thoracic injury directly accounts for 20-25% of deaths due to trauma; thoracic injury or its complications are a contributing factor in a further 25% of trauma deaths [24]. Approximately 85% of all thoracic trauma can be managed without surgical intervention; the mainstays of management are supplemental oxygen, intercostal drainage, good physiotherapy and pain control.
The primary survey of the ATLS course concentrates on the six immediately life-threatening injuries related to the airway and the chest. An additional six other major thoracic injuries constitute "the deadly dozen" of blunt thoracic trauma, which should be considered during the secondary survey (Table 1).
| Immediately life-threatening injuries | Potentially life-threatening injuries[23] |
| Airway obstruction | Simple pneumo/haemothorax |
| Tension pneumothorax | Aortic rupture |
| Open pneumothorax | Tracheobronchial rupture |
| Massive haemothorax | Pulmonary contusion |
| Flail chest | Blunt cardiac injury |
| Cardiac tamponade | Diaphragmatic rupture |
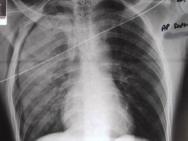
Aortic injuries cause or contribute to 15% of MVA fatalities[25]. Most patients with blunt aortic injury die before they reach hospital, and the vast majority will have major coexisting thoracic and extrathoracic injuries[26]. Widening of the mediastinum on an admission chest X-ray is the most common first sign of aortic injury[27]. Aortography is still the 'gold standard' diagnostic tool; however, contrast enhanced helical CT scanning also compares favourably with aortography[28]. A high index of suspicion may be required in the emergency room in order to diagnose thoracic aortic injury (TAI) as the initial chest X-ray may be normal and external evidence of injury may be minimal or absent; especially if the accident occurred with deployment of an airbag[29].
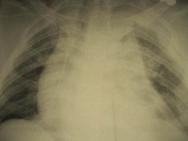
Blunt cardiac injury (BCI) refers to a spectrum of injuries ranging from simple electrocardiographic (ECG) changes to free rupture of the heart. The incidence of BCI is unknown, but clinically significant lesions are rare. There are few reliable clinical signs and symptoms that are specific for BCI. A diagnosis of BCI should be suspected in patients with an appropriate mechanism of injury, who show an inappropriate or abnormally poor cardiovascular response to their injury. At present, no single test or combination of tests has proven consistently reliable in detecting cardiac injury. Internet-based trauma management guidelines recommend a pragmatic treatment algorithm for patients suspected of having BCI[30]. An admission ECG should be performed on all patients in whom there is suspected BCI; and/or if the patient is haemodynamically unstable, an echocardiogram should be obtained. If an optimal transthoracic echocardiogram cannot be performed, then the patient should have a transesophageal echocardiogram. It is important to recognise that the presence of a sternal fracture does not predict the presence of BCI and, thus, does not necessarily indicate that monitoring should be performed. Neither creatinine phosphokinase with isoenzyme analysis nor measurement of circulating cardiac troponin T are useful in predicting which patients have or will have complications related to BCI. If the ECG is abnormal (abnormalities include arrhythmia, ischaemia, heart block, unexplained ST changes), then the patient should be admitted for continuous ECG monitoring for 24 to 48 hours. If the admission ECG is normal, the risk of having a BCI that requires treatment is insignificant [30].
Blunt tracheobronchial injuries constitute only a small fraction of admissions to trauma centres, as many patients die before they reach hospital [31][32]. Tracheobronchial injuries can be difficult to diagnose. Following tracheobronchial transection, peribronchial connective tissue may remain intact and allow distal lung ventilation. The presentation of thoracic tracheobronchial injury depends on whether the injury is confined to the mediastinum, or communicates with the pleural spaces. Injuries communicating with the pleural space usually present with haemoptysis, subcutaneous emphysema and a pneumothorax that persists despite adequate placement of chest tubes and a continuous air leak. If the injury is confined to the mediastinum then pneumomediastinum is usually present on X-ray.
The reported incidence of diaphragmatic rupture is between 0.8% and 1.6% of patients admitted to hospital with blunt trauma[33]. There is no single investigation that provides a reliable diagnosis of diaphragmatic rupture at presentation. Between 33% and 70% will be diagnosed on initial chest X-ray, but this is less reliable if the patient is intubated[34]. Diaphragmatic rupture can only be diagnosed on chest X-ray if stomach or bowel that has herniated is constricted by the torn diaphragm (the 'collar sign') or if the nasogastric tube is seen to lie above the diaphragm. The diagnosis of ruptured diaphragm is frequently missed in the acute phase because of the presence of hypovolaemic shock, respiratory insufficiency, visceral injuries or coma. Diagnostic delay can lead to long-term sequelae that can present a few days to many years after the injury. A high index of suspicion must be maintained, with careful evaluation of the domes of the diaphragm during each laparotomy for blunt trauma, or with appropriate radiological techniques.
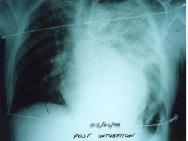
The most commonly injured intraabdominal organ following blunt trauma is the spleen. Laparotomy should be performed on haemodynamically unstable patients with suspected splenic injury. If the patient is stable, surgery may not be required as many injured spleens heal without surgical intervention. The occurrence of 'overwhelming post-splenectomy sepsis' in a minority of patients who have had a splenectomy for trauma, and a recognition that non-operative management of children with ruptured spleens reduces the requirement for blood transfusion and decreases the length of hospital stay compared with aggressive operative intervention, has led to an increased acceptance of non-operative management. This has now been expanded to include adult patients[35]. Adjunctive procedures, such as embolisation of splenic blood vessels have also been used to reduce the need for operative intervention[36].
Six factors have been identified that predict failure of conservative management for blunt splenic injury:
Haemodynamic instability
Grade of injury
Size of haemoperitoneum
Contrast blush on CT
Age older than 55 years
Pre-existent splenic disease[37].
Older patients and patients with injury to a diseased spleen were thought to be poor candidates for non-operative management; however, various studies have now been published that show neither factor should mandate operative treatment per se. Patients who are hypotensive at presentation and fail to stabilise with small volumes of resuscitation, or who develop recurrent hypovolaemic shock, should be operated on. If contrast is seen to 'blush' or pool on CT then angiography or surgery is required.
Any patient selected for non-operative management must be carefully assessed and reassessed by an experienced practitioner, with an operating theatre immediately available. Any deterioration requires operative intervention.
Approximately 85% of all patients with blunt hepatic trauma are stable. In this group, non-operative management significantly improves outcomes over operative management in terms of decreased abdominal infections, decreased transfusions, and decreased lengths of hospital stay [38]. For unstable patients, operative surgery is still the rule, with the 'damage control approach' now accepted as the standard of care[39]. Bleeding is controlled, often by use of perihepatic packing and the patient's time in the operating theatre is kept to a minimum. CT scanning is the mainstay of diagnosis for hepatic injuries after blunt trauma in the stable patient; the initial CT findings will help the trauma surgeon to determine the suitability for non-operative treatment.
Factors likely to lead to a failure of non-operative management of blunt liver injury have been identified as:
Haemodynamic instability
Grade of injury according to the American Association for the Surgery of Trauma Scale
Periportal tracking
Contrast pooling on CT scan [37]
Angio-embolisation should be reserved for haemodynamically stable patients. It enables embolisation of active bleeding and occlusion of pseudoaneurysms; however it is important that this is undertaken by experienced radiologists in an unit set up to use this technique.
Individuals subjected to high-speed deceleration in MVAs can experience rupture of intraabdominal hollow viscera. The mechanism of injury is thought to be compression of closed-loops of bowel by seat-belt restraints. Ecchymosis (bruising) may be seen across the torso in the distribution of the belt, but this finding is not universal. The reported incidence of bowel and mesenteric injuries after blunt abdominal trauma is approximately 1.3%[40]. The most pressing concern for surgeons treating blunt abdominal injury is occult HVI (i.e. injury that occurs and is clinically silent). These injuries can be difficult to identify on the non-invasive screening tests (CT and ultrasound), as there may be little associated bleeding. The increasing use of such tests to screen for intraabdominal injury and a reduction in the reliance on diagnostic peritoneal lavage increases the risk of missing these injuries. The accuracy of spiral CT is excellent for solid organ injury; however, the same cannot be said for HVI. Accurate and timely recognition can be difficult, and delay in diagnosis has been shown to be associated with significant morbidity and mortality [41]. In a recent review, several important observations were made:
Increasing grade of injury to a specific solid organ did not correlate with increasing rate of HVI
The higher the number of organs injured, the higher the incidence of HVI
Any combination of solid organ injury with pancreatic injury had a high rate of HVI.
When three solid organs were injured, HVI was 6.7 times more likely than when one solid organ was injured, and the presence of a pancreatic injury plus solid organ injury was associated with HVI in more than a third of patients. [42]. A high index of suspicion must be maintained and the surgeon must be prepared to repeat imaging techniques (CT scanning or diagnostic peritoneal lavage) or to proceed to operative intervention.
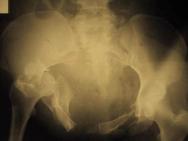
Fractures of the pelvis are increasingly recognised as a marker of severe injury, as the force required to disrupt the pelvic ring is substantial [24]. A high index of suspicion must be maintained for pelvic fracture, based on the history of the accident. All patients sustaining high-energy blunt injury should be assumed to have a pelvic fracture until proven otherwise[43].
There are many, confusing classification systems for pelvic fractures. Burgess et al have described a system based on the vector of force involved (lateral compression, anteroposterior compression, vertical shear, and combined mechanical injury) and a quantification of disruption from that force[44]. Their classification has been shown to correlate well with requirement for blood transfusion and mortality[44]. There are five places that a patient can lose a large volume of blood: externally into the environment, into the chest, abdomen, retroperitoneum and into muscle compartments. Severe bleeding leading to hypovolaemic shock is often a feature of severe pelvic fractures. Unstable pelvic fractures can bleed torrentially, and uncertainty about coexistent intraabominal injury may cause significant problems in identifying the source of blood loss. External fixation of an unstable pelvic fracture should be considered as early as possible. Returning the bony components of the pelvis into stable apposition enables haemostasis to occur and reducing the volume of a disrupted pelvis may also promote tamponade of bleeding within the pelvis [45]. Angiography and selective embolisation of bleeding vessels is an important adjunctive manoeuvre in major pelvic fracture. Assessment of the abdomen by diagnostic peritoneal lavage or CT for significant intraabdominal injury is vital [45].
Associated injury is common, and it has been stated that pelvic fractures cause problems for physicians who will not allow themselves to be diverted from the possibility of lethal visceral damage by the presence of broken bones, no matter how obvious these may appear[46]. Examination of the perineum, vagina and rectum is especially important in order not to miss open fractures or associated urethral injury.
Pelvic fracture may be associated with the following injuries (frequency):
Closed head injury (51%)
Long bone fracture (48%)
Peripheral nerve injury (26%)
Thoracic injury (20%)
Bladder (10%)
Spleen (10%)
Liver (7%)
Gastrointestinal tract (7%)
Kidney (7%)
Urethra (6%)
Mesentery (4%)
Diaphragm (2%).
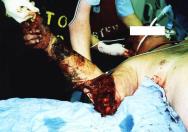
The optimal timing for long bone stabilisation in polytrauma patients has been debated for the last two decades. Reported benefits of early long bone stabilisation in polytrauma patients include increased patient mobilisation by eliminating the need for traction and decreased pulmonary morbidity. Although polytrauma patients who undergo long bone stabilisation within 48 hours of injury have no improvement in survival when compared with those receiving later stabilisation, there may be some patients who will have fewer complications. As there is no evidence that early stabilisation has any detrimental effect, the current recommendation is to perform early long bone stabilisation in polytrauma patients[47].
In patients with brain or chest injury, early long bone stabilisation appears to neither enhance nor worsen outcome. Current recommendations support management on an individual basis, with the important caveat to avoid any operative procedure that might lead to hypotension or hypoxia and cause a secondary insult[47]. The impressive appearance of an extremity injury must not distract practitioners from the priorities of resuscitation.
The first human fatality associated with a motor vehicle was a pedestrian killed in 1899 [5]; since then the patterns of injury from man's interaction with the motor car may have been somewhat modified by crash protection devices, such as helmets, seat belts and air bags, but injuries due to road traffic related trauma are worsening each year. Injury is the leading cause of death among young adults in the western world and trauma is imposing an increasingly severe burden on the health infrastructure of the developing world. The use of motor vehicles is growing worldwide; a particular concern in emerging nations where increasing urbanisation, overcrowding and scant regard for the 'rules of the road' are the norm.
A recognition of the typical patterns of injury coupled with a logical sequence for the initial assessment and management of trauma patients will contribute to reductions in mortality and morbidity; however, the most significant impact on reducing the worldwide burden of motor vehicle-related trauma will come from injury prevention programmes organised at societal and governmental levels.
1. Murray CJ, Lopez AD. Mortality by cause for eight regions of the world: Global Burden of Disease Study. Lancet 1997; 349(9061): 1269-76.
2. Murray CJ, Lopez AD. Alternative projections of mortality and disability by cause 1990-2020: Global Burden of Disease Study. Lancet 1997; 349(9064): 1498-504.
3. West JG, Trunkey DD, Lim RC. Systems of trauma care: a study in two countries. Arch Surg 1979; 114: 455-60.
4. McCoy GF, Johnstone RA, Nelson IW, Kenwright J, Duthie RB. Incidence and consequences of ejection in motor vehicle accidents. BMJ 1988; 297(6658): 1244-5.
5. Rivara FP, Grossman DC, Cummings P. Injury prevention. First of two parts. N Engl J Med 1997; 337(8): 543-48.
6. National Highway Traffic Safety Administration. Effectiveness of occupant protection systems and their use: third report to Congress. Washington DC: US Department of Transportation, December, 1996.
7. Evans L, Frick MC. Helmet effectiveness in preventing motorcycle driver and passenger fatalities. Accid Anal Prev 1988; 20(6): 447-58.
8. Johnson RM, McCarthy MC, Miller SF, Peoples JB. Craniofacial trauma in injured motorcyclists: the impact of helmet usage. J Trauma 1995; 38(6): 876-78.
9. Peek C, Braver ER, Shen H, Kraus JF. Lower extremity injuries from motorcycle crashes: a common cause of preventable injury. J Trauma 1994; 37(3): 358-64.
10. Thompson RS, Rivara FP, Thompson DC. A case-control study of the effectiveness of bicycle safety helmets. N Engl J Med 1989; 320(21): 1361-67.
11. van der Spuy JW. Trauma, alcohol and other substances. S Afr Med J 2000; 90(3): 244-46.
12. Schnyder U, Morgeli H, Nigg C, Klaghofer R, Renner N, Trentz O, Buddeberg C. Early psychological reactions to life-threatening injuries. Crit Care Med 2000; 28(1): 86-92.
13. Alexander DA. The psychiatric consequences of trauma. Hosp Med 2002; 63(1): 12-15.
14. Finfer SR, Cohen J. Severe traumatic brain injury. Resuscitation 2001; 48(1): 77-90.
15. MacKenzie EJ, Siegel JH, Shapiro S, Moody M, Smith RT. Functional recovery and medical costs of trauma: an analysis by type and severity of injury. J Trauma 1988; 28(3): 281-97.
16. Murray GD, Teasdale GM, Braakman R, Cohadon F, Dearden M, Iannotti F, et al. The European Brain Injury Consortium survey of head injuries. Acta Neurochir (Wien) 1999; 141(3): 223-36.
17. Thornhill S, Teasdale GM, Murray GD, McEwen J, Roy CW, Penny KI. Disability in young people and adults one year after head injury: prospective cohort study. BMJ 2000; 320(7250): 1631-35.
18. Baykaner K, Alp H, Ceviker N, Keskil S, Seckin Z. Observation of 95 patients with extradural hematoma and review of the literature. Surg Neurol 1988; 30(5): 339-41.
19. Burney RE, Maio RF, Maynard F, Karunas R. Incidence, characteristics, and outcome of spinal cord injury at trauma centers in North America. Arch Surg 1993; 128(5): 596-99.
20. McDonald JW, Sadowsky C. Spinal-cord injury. Lancet 2002; 359(9304): 417-25.
21. Bracken MB, Shepard MJ, Holford TR, Leo-Summers L, Aldrich EF, Fazl M, Fehlings M, et al. Administration of methylprednisolone for 24 or 48 hours or tirilazad mesylate for 48 hours in the treatment of acute spinal cord injury. Results of the Third National Acute Spinal Cord Injury Randomized Controlled Trial. National Acute Spinal Cord Injury Study. JAMA 1997; 277(20): 1597-604.
22. Fabian TC, Patton JH, Croce MA, Minard G, Kudsk KA, Pritchard FE. Blunt carotid injury. Importance of early diagnosis and anticoagulant therapy. Ann Surg 1996; 223(5): 513-22; discussion 522-25.
23. Kerwin AJ, Bynoe RP, Murray J, Hudson ER, Close TP, Gifford RR, et al. Liberalized screening for blunt carotid and vertebral artery injuries is justified. J Trauma 2001; 51(2): 308-14.
24. American College of Surgeons Committee on Trauma. Advanced Trauma Life Support Course. Chicago: American College of Surgeons, 1997.
25. Pretre R, Chilcott M. Blunt trauma to the heart and great vessels. N Engl J Med 1997; 336(9): 626-32.
26. Dosios TJ, Salemis N, Angouras D, Nonas E. Blunt and penetrating trauma of the thoracic aorta and aortic arch branches: an autopsy study. J Trauma 2000; 49(4): 696-703.
27. von Segesser LK, Fischer A, Vogt P, Turina M. Diagnosis and management of blunt great vessel trauma. J Card Surg 1997; 12(2 Suppl): 181-6; discussion 186-92.
28. Fishman JE. Imaging of blunt aortic and great vessel trauma. J Thorac Imaging 2000; 15(2): 97-103.
29. Dunn JA, Williams MG. Occult ascending aortic rupture in the presence of an air bag. Ann Thorac Surg 1996; 62(2): 577-78.
30. Pasquale MD, Nagy K, Clarke J. Practice Management Guidelines for Screening of Blunt Cardiac Injury. In: Practice management guidelines for trauma. The EAST Practice Management Guidelines Work Group: Eastern Association For The Surgery of Trauma, 1998. Available from URL: http://www.east.org/ [Accessed Sept 2002].
31. Bertelsen S, Howitz P. Injuries of the trachea and bronchi. Thorax 1972; 27(2): 188-94.
32. Ecker RR, Libertini RV, Rea WJ, Sugg WL, Webb WR. Injuries of the trachea and bronchi. Ann Thorac Surg 1971; 11(4): 289-98.
33. Shah R, Sabanathan S, Mearns AJ, Choudhury AK. Traumatic rupture of diaphragm. Ann Thorac Surg 1995; 60(5): 1444-49.
34. Simpson J, Lobo DN, Shah AB, Rowlands BJ. Traumatic diaphragmatic rupture: associated injuries and outcome. Ann R Coll Surg Engl 2000; 82(2): 97-100.
35. Schiffman MA. Nonoperative management of blunt abdominal trauma in pediatrics. Emerg Med Clin North Am 1989; 7(3): 519-35.
36. Davis KA, Fabian TC, Croce MA, Gavant ML, Flick PA, Minard G, et al. Improved success in nonoperative management of blunt splenic injuries: embolization of splenic artery pseudoaneurysms. J Trauma 1998; 44(6): 1008-13; discussion 1013-15.
37. Ochsner MG. Factors of failure for nonoperative management of blunt liver and splenic injuries. World J Surg 2001; 25(11): 1393-96.
38. Malhotra AK, Fabian TC, Croce MA, Gavin TJ, Kudsk KA, Minard G, Pritchard FE. Blunt hepatic injury: a paradigm shift from operative to nonoperative management in the 1990s. Ann Surg 2000; 231(6): 804-13.
39. Shapiro MB, Jenkins DH, Schwab CW, Rotondo MF. Damage control: collective review. J Trauma 2000; 49(5): 969-78.
40. Frick EJ, Pasquale MD, Cipolle MD. Small-bowel and mesentery injuries in blunt trauma. J Trauma 1999; 46(5): 920-26.
41. Fakhry SM, Brownstein M, Watts DD, Baker CC, Oller D. Relatively short diagnostic delays (<8 hours) produce morbidity and mortality in blunt small bowel injury: an analysis of time to operative intervention in 198 patients from a multicenter experience. J Trauma 2000; 48(3): 408-14; discussion 414-15.
42. Nance ML, Peden GW, Shapiro MB, Kauder DR, Rotondo MF, Schwab CW. Solid viscus injury predicts major hollow viscus injury in blunt abdominal trauma. J Trauma 1997; 43(4): 618-22; discussion 622-23.
43. Scalea TM, Burgess AR. Pelvic fractures In: Mattox KL, Feliciano DV, Moore EE, editors. Trauma (4th edition). New York: McGraw Hill, 2000; 807-38.
44. Burgess AR, Eastridge BJ, Young JW, Ellison TS, Ellison PS, Poka A, Bathon GH, Brumback RJ. Pelvic ring disruptions: effective classification system and treatment protocols. J Trauma 1990; 30(7): 848-56.
45. DiGiacomo JC, Bonadies JA, Cole FJ, Diebel L, Hoff WS, Holevar M, et al. Practice Management Guidelines for Hemorrhage in Pelvic Fracture. In: Practice management guidelines for trauma. The EAST Practice Management Guidelines Work Group: Eastern Association For the Surgery of Trauma, 2001. Available from URL: http://www.east.org/tpg/pelvis.pdf [Accessed Sept 2002].
46. Patterson BM. Pelvic ring injury and associated urologic trauma: an orthopaedic perspective. Semin Urol 1995; 13(1): 25-33.
47. Dunham CM, Bosse MJ, Clancy TV, Cole FJ Jr, Coles MJM, Knuth T, et al. Management Guidelines on the Optimal Timing of Long Bone Fracture Stabilization in Polytrauma Patients. In: Practice management guidelines for trauma. The EAST Practice Management Guidelines Work Group: Eastern Association For The Surgery of Trauma , 2002. Available from URL: http://www.east.org/tpg/longbone.pdf [Accessed Sept 2002].