

|
Author(s)
Wayne Naylor Wound
Management Research Nurse
|
Contents
|
|
Published:
Feb
2002
Last updated: Feb 2002 Revision: 1.0 |
Keywords: fungating wounds; sympton control; malodour; exudate; pain; bleeding.
For a significant minority of cancer patients the presence of a malodorous, exuding, necrotic skin lesion can be a constant physical reminder that their disease is both progressive and incurable.
Fungating wounds may develop from a primary skin tumour, an underlying tumour or metastatic disease. Sometimes a chronic wound may undergo malignant transformation to produce a fungating wound, although this is rare.
The most frequently reported symptoms related to a fungating wound are malodour, exudate, bleeding and pain.
Fungating malignant wounds are a distressing problem for a significant number of patients with advanced cancer. They may develop during the last few months of life or be present for a number of years. Fungating wounds rarely heal and often require palliative management. In treating patients with fungating wounds, the goal of care is to maintain or improve quality of life through symptom control. This article reviews the management of the four most common symptoms associated with fungating wounds: exudate, malodour, bleeding and pain.
Many cancer patients live with the knowledge that their disease is both progressive and incurable. For a significant minority of these people this reality may be present in the form of a malodorous, exuding, necrotic skin lesion, which can be a constant physical reminder of disease progression [1], [2]. These lesions are commonly known as 'fungating wounds', the term 'fungating' referring to a malignant process of both ulcerating and proliferative growth [3], [4]. Lesions that have a predominantly proliferative growth pattern may develop into a nodular 'fungus' or 'cauliflower' shaped lesion, whereas a lesion that is ulcerating will produce a wound with a crater-like appearance [5], [6]. Sometimes a lesion presents with a mixed appearance of both proliferating and ulcerating areas [7], [8].
A fungating wound may develop in the following ways:
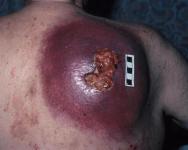
As a result of a primary skin tumour such as squamous cell carcinoma or melanoma (Figure 1).
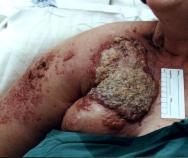
Through direct invasion of the structures of the skin by an underlying tumour, for example breast cancer (Figure 2), or haematological malignancy such as cutaneous T-cell lymphoma (mycosis fungoides).
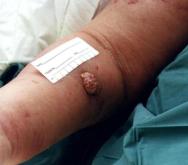
From metastatic spread of a distant tumour (Figure 3). Metastasis may occur along tissue planes, capillaries or lymph vessels.
Although rare, malignant changes may develop in chronic wounds (Marjolin's ulcer). This condition is most commonly associated with burn scar ulcers, but can develop in other chronic wounds such as pressure and venous stasis ulcers, as well as osteomyelitis [9], [10]. This type of malignancy usually occurs as an aggressive squamous cell carcinoma with a high rate of metastatic spread and local recurrence. In most cases, patients will be over the age of 50 and will have lived with the ulcer for between 25-40 years before any malignant changes occur. The condition is more common in women than men [11]. Signs of malignant change in a chronic wound include onset of pain or a change in the type or intensity of pain experienced, malodour and a change in the character, volume or appearance of exudate. A mass in the wound may be obvious, but a diagnosis of Marjolin's ulcer can only be confirmed following biopsy of the wound [9].
Fungating wounds tend to develop in elderly patients (>70 years) with metastatic cancer, who are in the terminal stages of their illness [12], [13]. Approximately 62% of fungating wounds will develop in the area of the breast and 24% in the head and neck area [14]; a fungating wound can, however, arise anywhere on the body. Although fungating wounds occur more frequently in people with advanced cancer, it is possible for people to live many years with a fungating wound if the disease is localised [5], [12], [15], [16]. For most patients, healing of the wound is unlikely unless there is a good response to anti-cancer therapy, such as radiotherapy or chemotherapy, or the wound can be surgically excised. Most fungating wounds will, however, continue to deteriorate over time [17], [18], [19] and commonly have a devastating effect on a person's physical and emotional well-being [5], [20], [21].
In treating patients with fungating wounds, the overall goals of nursing care are to promote patient comfort, confidence and a sense of well-being, to prevent isolation and maintain or improve a patient's quality of life (QOL) [12], [18], [22], [23]. Therefore, management is focused on controlling and/or eliminating the distressing symptoms associated with these wounds [7]. To achieve these goals accurate assessment is vital as a means of determining patient needs and evaluating the effectiveness of symptom management interventions [24].
A review of the literature has identified many physical, psychological and social problems associated with fungating wounds (Table 1). The most frequently reported wound symptoms are exudate, malodour, bleeding and pain.
| WOUND | PSYCHOSOCIAL |
| Anatomical location | Body image |
| Bleeding | Communication difficulties |
| Comfort of dressings | Cosmesis of dressing |
| Condition of surrounding skin | Denial |
| Condition of wound bed | Depression |
| Devitalised tissue | Embarrassment |
| Exudate management | Fear |
| Frequency of dressing changes | Guilt |
| Infection | Impact on family |
| Malodour | Information needs |
| Pain related to dressing changes | Restrictions due to dressing changes |
| Pruritis | Revulsion/disgust |
| Size/shape | Self-respect/self-esteem |
| Tunnelling/undermining | Sexuality |
| Wound pain | Shame |
| Social isolation | |
| Social support/resources |
Wound malodour is probably the most distressing symptom for patients [7], [19], [25], [26], and is caused by bacterial infection in devitalised tissue within the wound. This can also be devastating for the patient's family and caregivers, as wound malodour may be constantly detectable and can trigger gagging and vomiting reflexes [15], [27], [28]. The presence of a pervasive malodour can lead to embarrassment, disgust, depression and social isolation and may have a detrimental effect on sexual expression causing relationship problems [18], [19], [21], [28].
Fungating wounds often produce excessive amounts of exudate which can be difficult to manage [20], [21]. Increased permeability of blood vessels within the tumour and secretion of vascular permeability factor by tumour cells are the most likely causes of high exudate levels [12]; exudate production may also be increased if infection is present. Large volumes of uncontrolled exudate may cause leakage from the dressing [3] and staining of the patient's clothes [29], [30], [31]. This again can cause significant psychosocial problems for patients and carers. Effective management of wound exudate is likely to increase both patient confidence and comfort.
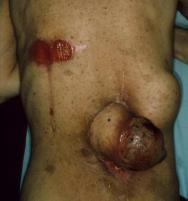
There are a number of mechanisms that can cause fungating wounds to be painful. The tumour, for example, may be pressing on nerves and blood vessels (Figure 4), or there may be exposure of the dermis [32]. Patients often describe their pain as a superficial stinging or painful ulceration [5]. Painful procedures such as the use of an inappropriate cleansing technique or removal of a dressing which adheres to the wound bed may also impact on the pain experienced by the patient [21].
Wound bleeding is common in fungating wounds. This occurs because malignant cells erode blood vessels, and may be compounded by decreased platelet function within the tumour. Profuse, spontaneous bleeding can be distressing for both patients and their carers [12], while damage to fragile tissues during dressing changes may exacerbate bleeding (Figure 5).
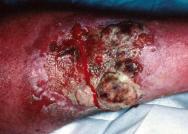
The position of the wound may be a source of embarrassment and distress for the patient especially if it is in an area such as the breast or genitals, or an area highly visible to others, for example, on the face [20]. Appropriate wound management that attends to the patient's outward appearance can have a very positive effect on self-esteem and emotional distress [12], [18]. Many patients can only continue an active social life if a socially acceptable dressing is available [31]. In some cases this may be achieved by devising a cosmetically acceptable dressing regime, for example, one that not only effectively manages odour and exudate, but also restores body symmetry. This should be carried out in an effort to boost patient confidence and improve their ability to socialise [31], [33].
Debridement removes necrotic tissue and bacteria and is the primary treatment for malodorous fungating wounds. Surgical or sharp debridement, however, is not recommended because of the increased tendency of these wounds to bleed. Autolytic or enzymatic debridement is the preferred method, providing this does not significantly increase exudate production [34]. Antibiotic therapy can also be effective if this destroys the bacteria responsible for malodour [35], [36], [37], the most common treatment being metronidazole. This may be given systemically, but side effects such as nausea, neuropathy and alcohol intolerance may affect patient acceptability [38]. A poor blood supply to the wound may further reduce the effectiveness of systemic treatment [39]. Topical preparations of metronidazole gel (e.g. Metrotop®, Anabact®) have been used successfully [40], [41]. This gel is usually applied once daily for five to seven days but may need to be repeated more often to keep malodour under control.
The use of activated charcoal dressings can have an immediate effect on wound malodour. These include plain activated charcoal cloth (e.g. CliniSorb®), or charcoal cloth combined with other dressing materials (e.g. CarboFlex®, Lyofoam C®) or impregnated with silver (e.g. Actisorb Silver 220®). Activated charcoal attracts and binds the volatile odour causing molecules, preventing their escape from the local wound area [42].
Sugar paste and honey have recently come back into use, mainly due to the emergence of many antibiotic resistant strains of bacteria, and both have antibacterial and debriding properties. The high sugar content of these products produces a hyperosmotic wound environment that inhibits bacterial growth and assists in wound debridement [43], [44]. Honey may also contain bacteriocidal hydrogen peroxide, which is slowly released as the honey is diluted in wound exudate, while specific types of honey may have plant derived antibacterial properties [45], [46], [47].
Occlusive dressings may help by containing wound malodour, while daily dressing changes with the correct disposal of soiled dressings, can help prevent build-up of stale exudate. Deodorisers may mask the odour and products such as essential oils, environmental air filters or commercial deodorisers may be helpful [48]. However, on occasions these products, in particular commercial deodorisers, may make the odour worse or cause unpleasant associations with smells.
A variety of dressings have been evaluated for the management of exudate from fungating wounds with varying levels of success [49], [50]. Where exudate is low, wounds should be managed with dressings that have a low absorbency so as not to dry out the wound, for example hydrocolloids, semi-permeable films and low adherent absorbent dressings [21]. However, care should be taken with the latter as they may stick to the wound base if this dries out too much.
More commonly fungating wounds produce moderate to high levels of exudate and it is important to choose a dressing that will absorb excess exudate, but still maintain a moist wound environment. Suitable dressings for wounds with high exudate levels include alginate and hydrofibre dressings, foam dressings and non-adherent wound contact layers, such as soft silicone (Mepitel®) or knitted viscose (NA Ultra®) with a secondary absorbent pad [5], [20]. For wounds with a small opening and high exudate, a stoma appliance or wound manager can be used [21].
Assessment of pain is vital as this will enable the clinician to understand the type of pain the patient is experiencing and determine the most appropriate treatment. Analgesic drugs should be prescribed using the World Health Organisation (WHO) guidelines for the control of cancer pain [51] and in accordance with local prescribing guidelines. It may also be useful to give the patient a pre-medication before dressing changes or a booster dose of their usual opiate, for example Oramorph® if the patient is on MST®. If dressing changes are very painful it may be worth using nitrous oxide (Entonox®) gas during the procedure [52].
To help prevent pain during dressing changes, non-adherent dressings should be used. Maintaining the wound in a moist environment will not only reduce dressing adherence but will also protect exposed nerve endings [18], [53]. If pain cannot be controlled at dressing changes then it may be worth trying a product that requires less frequent changes. Irrigation of the wound with warm saline rather than cleaning with a gauze swab will, in some cases, reduce pain [54]. Complementary therapies can play an important part in pain management; therapies such as relaxation, distraction or visualisation may help anxious and stressed patients who will have a heightened response to pain [55], [56].
Topical opioids are an interesting alternative form of pain control that can be used for painful ulcerating wounds. Although research is limited, a number of published case studies and theoretical papers support the use of topical opioids to reduce wound pain [57], [58], [59], [60], [61]. Morphine and diamorphine are most commonly used, mixed with a hydrogel (about 1 mg of morphine to 1 g of hydrogel for 0.08 to 0.1% mixture). This is usually applied to the wound once daily although some patients have used it on an 'as required' basis.
Chemotherapy, radiotherapy, hormone therapy or a combination of these anti-cancer therapies may help to shrink the wound by destroying malignant cells, reducing pressure on nerves and other structures and decreasing the area of exposed tissue [12], [18], [62]. This may have a significant effect on the level of pain experienced by the patient.
Preventative measures are important to reduce the risk of bleeding. Using non-adherent dressings that maintain a moist environment, and cleansing by irrigation rather than swabbing, will reduce the risk of trauma and subsequent bleeding. Oral antifibrinolytics such as tranexamic acid (Cyklokapron®) may also help [20].
For wounds that are actively bleeding, there are a number of strategies available. Sucralfate paste or an alginate may be applied to wounds with a small amount of bleeding [63], [64]. Alginates should, however, be used with caution in fragile tumours as they may cause bleeding [65]. As an alternative, haemostatic surgical sponges, such as Spongostan® or Oxycell®, can be used as these promote rapid haemostasis and can be left in place and covered with an appropriate dressing. Topical adrenaline or tranexamic acid can also be applied, but they should only be used under medical supervision and caution is advised as adrenaline may cause ischaemic necrosis due to local vasoconstriction [61]. Excessive, uncontrolled bleeding may need referral to a vascular surgeon for cautery or ligation.
Fungating wounds can present a major problem for both patients and healthcare professionals. Not only do they signify progressive and life threatening disease, but also significantly affect a patient's QOL by causing unpleasant and difficult to manage symptoms.
The cornerstone of effective symptom management is accurate assessment. In Part Two, the development of a new symptom assessment tool is reviewed, which allows patients to rate the severity of symptoms and problems they are experiencing, together with the level of interference their symptoms and associated problems are having on their lives.
1. Mortimer PS. Management of skin problems: medical aspects In: Doyle D, Hanks GWC, MacDonald N, editors. Oxford Textbook of Palliative Medicine (2nd edition). Oxford: Oxford University Press, 1998; 617-27.
2. Englund F. Wound management in palliative care. RCN Contact 1993; Winter: 2-3.
3. Grocott P. The palliative management of fungating malignant wounds. J Wound Care 1995; 4(5): 240-2.
4. Bycroft L. Care of a handicapped woman with metastatic breast cancer. Br J Nurs 1994; 3(3): 126-8, 130-3.
5. Grocott P. The management of fungating wounds. J Wound Care 1999; 8(5): 232-4.
6. Collier M. The assessment of patients with malignant fungating wounds - a holistic approach: Part 1. Nurs Times 1997; 93(44): suppl 1-4.
7. Young T. The challenge of managing fungating wounds. Community Nurse 1997; 3(9): 41-4.
8. Carville K. Caring for cancerous wounds in the community. J Wound Care 1995; 4(2): 66-8.
9. Malheiro E, Pinto A, Choupina M, Barroso L, Reis J, Amarabte J. Marjolin ulcer of the scalp: case report and literature review. Annals of Burns and Fire Disasters (online) 2001; 14(1):
10. Esther RJ, Lamps L, Schwartz HS. Marjolin ulcers: secondary carcinomas in chronic wounds. J Southern Orthopaedic Assoc 1999; 8(3): 181-87.
11. Hill BB, Sloan DA, Lee EY, McGrath PC, Kenady DE. Marjolin's ulcer of the foot caused by nonburn trauma. South Med J 1996; 89(7): Available from URL: http://www.sma.org/smj/96jul11.htm [Accessed 28 November 2001].
12. Haisfield-Wolfe ME, Rund C. Malignant cutaneous wounds: a management protocol. Ostomy Wound Manage 1997; 43(1): 56-60, 62, 64-6.
13. Ivetic O, Lyne PA. Fungating and ulcerating malignant lesions: a review of the literature. J Adv Nurs 1990; 15(1): 83-8.
14. Thomas S. Current Practices in the Management of Fungating Lesions and Radiation Damaged Skin. Bridgend, Mid Glamorgan: The Surgical Materials Testing Laboratory, 1992.
15. Collier M. The assessment of patients with malignant fungating wounds - a holistic approach: Part 2. Nurs Times 1997; 93(46): suppl 1-4.
16. Goodman M, Hilderley LJ, Purl S. Integumentary and mucous membrane alterations In: Groenwald SL, Hansen Frogge M, Goodman M, Henk Yarbro C, editors. Cancer Nursing: Principles and Practice (4th edition). Massachusetts: Jones and Bartlett, 1997; 768-822.
17. Bale S, Jones V. Wound Care Nursing. A Patient Centred Approach. London: Baillere Tindall, 1997.
18. Hallett A. Fungating wounds. Nurs Times 1995; 91(47): 81-83.
19. Haughton W, Young T. Common problems in wound care: malodorous wounds. Br J Nurs 1995; 4(16): 959-60, 962-3.
20. Pudner R. The management of patients with fungating or malignant wound. J Community Nursing (online) 1998; 12(9): Available from URL: http://www.jcn.co.uk/backiss.asp?YearNum=1998&MonthNum=09&ArticleID=82 [Accessed 18 February 2000].
21. Jones M, Davey J, Champion M. Dressing wounds. Nurs Stand 1998; 12(39): 47-52; quiz 55-6.
22. Laverty D, Cooper J, Soady S. Wound management. In: Mallet J, Dougherty L, editors. The Royal Marsden Hospital Manual of Clinical Nursing Procedures (5th edition). Oxford: Blackwell Scientific Ltd, 2000; 681-710.
23. Ingham J, Portenoy RK. The measurement of pain and other symptoms In: Doyle SL, Hanks GWC, MacDonald N, editors. Oxford Textbook of Palliative Medicine (2nd edition). Oxford: Oxford University Press, 1998; 203-19.
24. Samarel N, Leddy SK, Greco K, Cooley ME, Torres SC, Tulman L, Fawcett J. Development and testing of the symptom experience scale. J Pain Symptom Manage 1996; 12(4): 221-8.
25. Price E. Wound care. The stigma of smell. Nurs Times 1996; 92(20): 70-2.
26. Fairburn K. A challenge that requires further research: management of fungating breast lesions. Professional Nurse 1994; 9(4): 272-77.
27. Gallagher J. Management of cutaneous symptoms. Semin Oncol Nurs 1995; 11(4): 239-47.
28. Van Toller S. Invisible wounds: the effects of skin ulcer malodours. J Wound Care 1994; 3(2): 103-05.
29. Davis V. Goal-setting aids care. Nurs Times 1995; 91(39): 72, 74, 76.
30. Boardman M, Mellor K, Neville B. Treating a patient with a heavily exuding malodorous fungating ulcer. J Wound Care 1993; 2(2): 74-76.
31. Grocott P. Wound care - practical changes. Nurs Times 1993; 89(7): 64, 66, 68 passim.
32. Manning MP. Metastasis to skin. Semin Oncol Nurs 1998; 14(3): 240-3.
33. Saunders S. Mutual support. Nurs Times 1997; 93(32): 76-82.
34. Grocott P. The palliative management of fungating wounds. In: Educational booklet Huntingdon: Wound Care Society, 2001.
35. Bower M, Stein R, Evans TR, Hedley A, Pert P, Coombes RC. A double-blind study of the efficacy of metronidazole gel in the treatment of malodorous fungating tumours. Eur J Cancer 1992; 28A(4-5): 888-9.
36. Newman V, Allwood M, Oakes RA. The use of metronidazole gel to control the smell of malodorous lesions. Palliative Medicine 1989; 34: 303-05.
37. Ashford RF, Plant GT, Maher J, Pickering D, Coe MA, Drury A, Goold MA, Teare EL. Metronidazole in smelly tumours. Lancet 1980; 1(8173): 874-5.
38. Hampton JP. The use of metronidazole in the treatment of malodorous wounds. J Wound Care 1996; 5(9): 421-26.
39. Thomas S, Fisher B, Fram PJ, Waring MJ. Odour-absorbing dressings. J Wound Care 1998; 7(5): 246-50.
40. Gilchrist B. Wound infection In: Miller M, Glover D, editors. Wound Management Theory and Practice. London: Nursing Times Books, 1999; 96-106.
41. Cutting K. Wounds and infection. In: Educational leaflet Huntingdon: Wound Care Society, 1998.
42. Williams C. Clinisorb activated charcoal dressing for odour control. Br J Nurs 1999; 8(15): 1016-19.
43. Edwards J. Wound management (2): managing malodorous wounds. J Community Nursing (online) 2000; 14(4): Available from URL: http://www.jcn.co.uk/backiss.asp?YearNum=2000&MonthNum=04&ArticleID=136 [Accessed 2 July 2001].
44. Cooper R, Molan P. The use of honey as an antiseptic in managing Pseudomonas infection. J Wound Care 1999; 8(4): 161-4.
45. Molan PC. The role of honey in the management of wounds. J Wound Care 1999; 8(8): 415-8.
46. Morgan DA. Formulary of Wound Management Products: A Guide for Healthcare Staff (8th edition). Surrey, UK: Euromed Communciations Ltd, 2000.
47. Molan PC. Honey as a topical antibacterial agent for treatment of infected wounds. World Wide Wounds (online) 2001; Available from URL: http://www.worldwidewounds.com/2001/november/Molan/honey-as-topical-agent.html [Accessed February 2002].
48. Naylor W, Laverty D, Mallett J. The Royal Marsden Hospital Handbook of Wound Management in Cancer Care. Oxford: Blackwell Science Ltd, 2001.
49. Naylor W. Using a new foam dressing in the care of fungating wounds. Br J Nursing 2001; 10(6): Suppl S24-S30.
50. Grocott P. Exudate management in fungating wounds. J Wound Care 1998; 7(9): 445-8.
51. World Health Organisation. Cancer Pain Relief (2nd edition). Geneva: WHO, 1996.
52. Travis S. Entonox administration In: Mallet J, Dougherty L, editors. The Royal Marsden Hospital Manual of Clinical Nursing Procedures (5th edition). Oxford: Blackwell Science Ltd, 2000; 261-65.
53. Emflorgo CA. The assessment and treatment of wound pain. J Wound Care 1999; 8(8): 384-5.
54. Hollinworth H. Wound care - less pain, more gain. Nurs Times 1997; 93(46): 89-91.
55. Ryman L, Rankin-Box D. Relaxation and visualisation In: Rankin-Box J, editor. The Nurse's Handbook of Complementary Therapies (2nd edition). London: Balliere Tindall, 2001; 251-58.
56. Downing J. Pain in the Patient with Cancer, Nursing Times Clinical Monographs No5. London: NT Books, 1999.
57. Stein C. The control of pain in peripheral tissue by opioids. N Engl J Med 1995; 332(25): 1685-90.
58. Twillman RK, Long TD, Cathers TA, Mueller DW. Treatment of painful skin ulcers with topical opioids. J Pain Symptom Manage 1999; 17(4): 288-92.
59. Back IN, Finlay I. Analgesic effect of topical opioids on painful skin ulcers. J Pain Symptom Manage 1995; 10(7): 493.
60. Krajnik M, Zylicz Z. Topical morphine for cutaneous cancer pain. Palliat Med 1997; 11(4): 325.
61. Grocott P. Palliative management of fungating malignant wounds. J Community Nursing (online) 2000; 14(3): Available from URL: http://www.jcn.co.uk/backiss.asp?YearNum=2000&MonthNum=03&ArticleID=221 [Accessed 15 June 2001].
62. Miller C. Management of skin problems: nursing aspects In: Doyle D, Hanks GWC, MacDonald N, editors. Oxford Textbook of Palliative Medicine (2nd edition). Oxford: Oxford University Press, 1998; 642-56.
63. Thomas S, Vowden K, Newton H. Controlling bleeding in fragile fungating wounds. J Wound Care 1998; 7(3): 154.
64. Emflorgo CA. Controlling bleeding in fungating wounds. J Wound Care 1998; 7(5): 235.
65. Grocott P. Controlling bleeding in fragile fungating tumours. J Wound Care 1998; 7(7): 342.