

Dr. Lawrence O Kollenberg [gcfc@ipa.net], Director Garland County Foot Clinic |
|
Submitted: 14 April 1998 Published: 29th June 1998 Edition: 1.0 |
Clinical evidence is used to illustrate how three commonly utilized antibiotics can be impregnated into collagen in order to assist with achieving resolution of infection in deep soft tissue or osteomyelitis infections of the feet.
Today's health care practitioner has the burden of providing cost effective care. Infections are no exception. Today we all try to manage our patients with a minimal number of hospital stays. This case report shows a possible new method of drug delivery of antibiotics in serious, significant infections including deep space soft tissue or osteomyelitis infections of the feet that may lead to minimizing need for inpatient care.
As clinicians treat infections there exist certain basic concerns on all patients. These include such problems as general health status (e.g. diabetes, peripheral vascular disease, collagen vascular disorders). In arterial ischemic diseases, the lack of blood flow to tissue results in compromise of delivery of macrophages and nutrients to allow cells to fight infections. Similarly antibiotic delivery in the bloodstream and thus to the tissues is compromised.
The protocols listed in this article are designed for clinicians to test these results on their own patients to see if in fact this new drug delivery system can assist in resolving the enigma of amputation and perhaps shorter length of stay in these cases.
The collagen molecule is composed of two alpha and one alpha2 chains wrapped in a triple helix with three left handed helical alpha chains twisted into a right handed super helix structure.[3,4] The alpha chains are similar in structure and contain approximately 1000 amino acid residues where commonly every third molecule is glycine. The other amino acids are either leucine and proline, or hydroxyleucine and hydroxyproline.[4] To maintain this structure, hydrogen bonds and disulfide bonds must remain intact or it results in a non-functional protein.[5] This triple helix arranged collagen is known as fibrillar collagen.[1]
Collagen has been reported as a well tolerated and completely biodegradable material. In addition, collagen has been utilized as a carrier system for occular application, involving pilocarpine and macrolide antibiotics.[6,7,8] Collagen has been shown to be a drug carrier vehicle for lipophillic drugs including tretinoin and lidocaine.[4] Collagen, when combined with chitosan has acted as a transdermal delivery system for delivering calcium channel blocker, and beta blocker drugs.[9] The releasing system of collagen has been shown to be sustained over time (approximately 2.5 days) in a linear fashion.[10] Collagen sheets with impregnated gentamycin have been used to treat chronic osteomyelitis.[11] During the course this study, the systemic absorption of gentamycin after the collagen sponge was implanted into patients, was found to be minimal (if present at all). The mechanism elucidated for this process shows that the collagen triple helix contains about 17% charged amino acid residues which are uniformly distributed over its entire length.[10] Under physiological conditions, the overall net charge is neutral. With specific chemical reactions, the collagen molecule can be modified to yield a net negative, or a net positive charge. This results in an enhanced retention of the oppositely charged drugs by binding interactions.[10]
When collagen sponge is combined with liposome encapsulated antibiotics, the duration of time for release of the antibiotic has been reported to be up to three times greater than that of collagen sponge alone.[12] Polymixin B and Amikacin have been shown in other laboratory experiments to have significant sustained release action against Pseudomonas Aeruginosa when attached to type I collagen.[13] There exists significant data that collagen when applied topically to open wounds may accelerate the healing capacity of wounds.[14] Furthermore, this study demonstrated that gentamycin impregnated collagen sponge shows up to 600 times MIC as compared to methyl methacrylate beads at 300 times MIC. Methyl methacrylate beads are reported to be sporadic in their release rate, whereas there has been significant evidence brought to bear that there may exist a constant linear diffusion rate of collagen giving a sustained release of many drugs [6-13]
The literature suggests that type I collagen, in a triple helix arrangement, may give a steady sustained release of antimicrobial agents in infectious disease. This report will review the clinical application of this in 12 patients with active, clinically significant infections involving deep soft tissues or bone.
| TABLE 1: Initial evaluation | |||||||
| No. | Age Range | ABI<7 | NIDDM | IDDM | osteo-myelitis | soft tissue infection only | |
| Male | 5 | 68-82 | 3 | 2 | 2 | 3 | 2 |
| Female | 7 | 63-98 | 5 | 2 | 3 | 5 | 2 |
| TABLE 2: Blood Analysis |
|
There were no patient admissions or IV administration of antibiotics during the course of care. Outside home health agency nurses were trained in this protocol to assist with patient home bound care on a daily basis. Nurses also monitored patient compliance for diabetes checks, antibiotics being administered, etc.
All the patients were initially treated with oral antibiotics. Empirical choice was based upon patient history, duration of infection, type of infection, (i.e. soft tissue or bone). Oral agents included Cephalexin 2 grams (split dosing four times daily) or Clindamycin 900mg (split dosing three times daily)
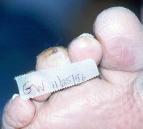
At the initial clinic visit, all infected areas were carefully evaluated and prepped with chloroxylenol 3.0%, cocamidopropyl PG-dimonium chloride phosphate 3.0% (PCMX 3.0%) for two minutes, following University of Minnesota transplant pre-operative surgical prep protocol.[15] When open and draining with apparent deep portal ulceration, wounds were probed to determine if infection traveled to the bone. (Figure 1)
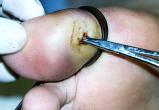
If the lesion had not been probed prior to debridement, the sites were surgically traced at the time of initial surgical debridement, to determine if infection was in bone. Plain radiograph, bone, and soft tissue changes were used to aid diagnosis.
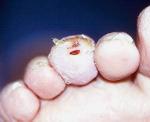
Deep soft tissue specimen or bone specimen (Figure 2) was obtained for culture and sensitivity analysis. Upon completion of surgical debridement, wounds were packed with type I bovine collagen mixed with gentamycin (Figure 3) followed by secondary sterile dressing.
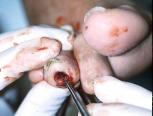
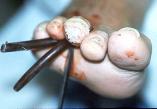
Initially, if the lesion involved distal toes, then 5cm x 5cm gauze was rolled and placed into sulcus of toe at the initial dressing (Figure 4) to assist with depressurizing the digit. All 12 patients were placed into surgical shoe, which was modified to for depressurization of the affected area and to allow immediate limited ambulation (meal table and bathroom).
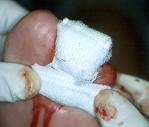
Patients were given clear and consistent instructions at the
first clinic attendance, as follows:
All patients attended clinic at least twice the first week and then weekly for serial evaluations until the infection was resolved and wounds were closed. All laboratory reports were re-evaluated at the second visit. When bacterial organisms were determined to be resistant to gentamycin or the oral agent, then a change in antibiotics was made (Table 3).
| TABLE 3: Organisms from culture | ||
| Organisms isolated | Number of Isolates | Gentamycin Resistant |
| Staphylococcus Aureus | 6 | 2 |
| Methicillin Resistant Staph. Aureus | 4 | 3 |
| Staphylococcus Epidermidis | 5 | 2 |
| Enterococcus | 3 | 3 |
| Pseudomonas Aureginosa | 3 | 0 |
| Klebsiella Pneumonia | 2 | 0 |
| Escherechia Coli | 1 | 0 |
| Proteus Miralbis | 2 | 0 |
| 5 patients had 1 isolate 3 patients had 2 isolates 2 patients had 3 isolates 1 patient had 4 isolates 1 patient had 5 isolates |
||
Three of the patients received Clindamycin impregnated into type I bovine collagen and three of the patients received Vancomycin impregnated into type I bovine collagen. If resistance was reported to initial oral agents, selected patients oral agent was changed based upon sensitivity results from culture report. Patients remained on oral agent until infection was localized.
All patients had repeat of CBC with differential, ESR, CRP, every two weeks until results returned to normal. Serial plain radiographs were utilized to follow osteomyelitis cases until clinical and radiographic resolution of osteomyelitis was determined. In the event of premature closure, then infection site was re-opened (Figure 5and 6).
Patients or home health nurses were instructed on dressings application as follows:
The gauze cover was applied because many patients found that the film dressing would roll or stick to clothing; the gauze reduced the incidence of interference. Dressings were applied daily, because patients were unwilling to accept alternate-day dressings.
| Table 4: Products and Manufacturers | ||
| Generic Name | Proprietary Name | Manufacturer |
| Gentamycin | Garamycin | Schering Plough |
| Clindamycin | Cleocin | Upjohn |
| Vancomycin | Vancocin | Eli Lilly |
| PCMX 3.0% | Technicare | Care Tech Labs |
| Surgical Shoe | Darco Shoe | Darco, Inc |
| Crest Pads | n/a | Stein Foot Specialties |
| Cut-out Pads | n/a | Stein Foot Specialties |
| Type I Bovine Collagen | Medifil | BioCore, Inc |
| Cephalexin | Keflex | Eli Lilly |
| Ofloxacin | Floxin | McNeil |
| Doxycycline | Vibramycin | Pfizer |
| Sulfamethoxazole | Bactrim DS | Roche Labs |
| Trimethoprim | ||
| Gauze | Kling | Johnson & Johnson |
| Specialty Absorptive Pads | Ex-U-Dry | Ex-U-Dry, Inc. |
| Foam | Hydrosorb | Convatec |
| Adhesive Film Dressing | Opsite | Smith and Nephew |
| Gauze Netting | Spandage | Medi-Tech International |
| Sterile Saline | for irrigation | Baxter Healthcare |
The combination of antibiotic impregnated into the collagen is generically described below and specifically listed in Table 5 for individual antibiotics. A total of 15cc of type I bovine particles were poured into a sterile container. The antibiotic was then measured in a sterile syringe and squirted slowly directly on the particles in the container. With a sterile tongue depressor blade, the mixture was stirred until non-lumpy paste was achieved. The container utilized is a light, air tight container. (Note: the author is unaware of any shelf life data of this mixture.) The patients were instructed to storethe mixture in a dark, non-humid cabinet/closet, such as linen closet at home.
| Table 5: Mixing Table |
| Gentamycin: Mix 4cc (160mg) of gentamycin with 15cc mature lyophilized type I bovine collagen particles Yields 10mg of gentamycin/cc of collagen particles. |
| Clindamycin: Mix 4cc (600mg) of clindamycin phosphate with 10cc of mature lyophilized type I bovine collagen particles Yields 60mg of clindamycin/cc of collagen particles. |
| Vancomycin: Dissolve 1 g of vancomycin powder with 10cc of a local anesthetic (lidocaine). Then mix the solution with 15cc of mature lyophilized type I bovine collagen particles Yields 66mg of vancomycin/cc of collagen particles. |
At the initial visit, patients were fitted for their surgical shoes which were later modified with additional pads or toe crests to depressurize the infected location. After the initial debridement, infected wounds were intentially left open to drain. Foams and specialty absorptive pads were applied to keep external contamination to a minimum.
Oral agents utilized were changed based upon the sensitivity results of tissue cultures. Additional antibiotics selected included Ofloxacin, sulfamethoxazole trimethoprim, doxycycline. Oral agents were continued until infectious disease was localized.
Oral agents were continued, discontinued, or changed based upon the following criteria:
The oral agents utilized in all cases were discontinued after three weeks. The standard of care in osteomyelitis is a minimum of six weeks; the remainder of the infectious disease resolution was thus the topical treatment of the antibiotic impregnated into the type I bovine collagen.
Of the 12 patients initially started on gentamycin in type I
collagen, six remained with this form of topical therapy until all
appearance of infection was resolved clinically and the wound areas
were 100% re-epithelialized. Serial radiographic evidence was
followed clinically with plain x-rays obtained every three weeks
for all osteomyelitis patients. Of the six patients to which
culture biopsy revealed resistance to gentamycin, three of the
patients were started onto Clindamycin impregnated into type I
bovine collagen and three patients were started on Vancomycin
impregnated into type I bovine collagen. (Table
5)
Patient resolution to complete healing ranged from 35 to 162
days.
Patient VA was referred as an alternative to amputation by his primary care physician. His ABI was reported as 0.41 with toe pressures of 60mm of Hg. His initial care consisted of avulsion of his second toenail, debridement of necrotic tissue, curretage of osteomyelitic bone with bone specimen for tissue culture report. He had a polymicrobial infection. He was changed to collagen/clindamycin on the 4th day and the wound had completely resolved in approximately 90 days (Figures 7-27).
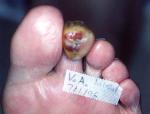
The patient had been treated at a local diabetes treatment center without success. Recommendation from orthopedic specialist in foot and ankle was to admit to hospital for IV antibiotics and schedule for amputation. ABI had been reported to be 0.41 with toe pressure reported as approximately 60mm Hg. It can be seen in Figure 7 that the toenail looks non-viable. There is mild generalized edema, without gross severe systemic cellulitis.
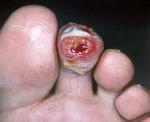
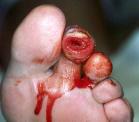
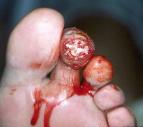
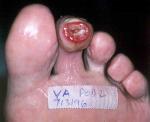
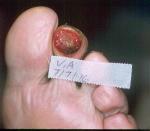
Home health nurses had been asked to change from gentamycin to clindamycin in collagen, 48 hours prior to this visit. Necrotic tissue was debrided at this visit; clindamycin in collagen and other orders to be continued.
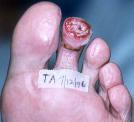
On day 13 (see (see Figure 13), minimal necrotic tissue is noted. Perimeter of wound shows good granular bed that is present. Collagen lattice is present in mid-central portion of wound bed.
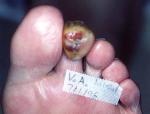
On day 18 (see Figure 14), the digit showed minimal edema and erythema. There was still a dusky appearance to the toe, but the wound continued to show improvement in size, shape, and appearance. The toe is shown post debridement. The granular tissue continues to develop. Margin depth was getting narrower. Appearance of keratin development to perimeter as wound gets smaller and contracts inward.
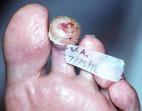
Day 25 (Figure 15), the wound has developed a hyperkeratotic. There is minimal maceration with collagen lattice in the central portion of wound bed.
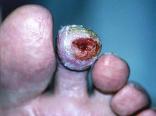
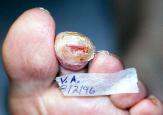
Figure 17 shows the wound on day 32, pre-debridement. The antibiotic collagen mixture has liquified but has not been absorbed by the external secondary dressing. Hyperkeratosis build-up, as seen in previous pictures, is significantly lessened this time.
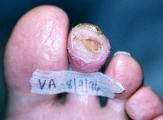
After debridement (Figure 18), erythema is minimized. Digit shows minimal bulby aspects to it. No local edema is visible. Change in position of shoe buttress pad was made this visit.
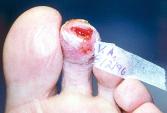
Figure 19 on day 39 shows the wound pre-debridement. The hyperkeratotic rim is significant in spite of attempts to depressurize digit. A change in position of the shoe buttress pad was made at this visit.
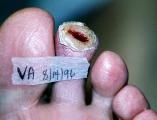
It can be seen in Figure 20, from day 53, that the toe shows no erythema, edema, undermining or tunneling. The wound is continuing to shrink.

On day 74, (Figure 21), the wound is epithelialized. The digit shows complete resolution of infectious process.
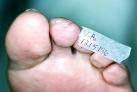
At a follow-up visit 10 weeks later (Figure 22), the wound site is 100% epithelialized. The toe shows minimal scarification and the skin is soft, supple, pliable.
Prior antibiotic usage had included Ciprofloxacin. Her culture report for the gram negative organisms was extended out to specifically test for the Clindamycin she was started on. Although the polymicrobial infection was sensitive to it, she developed diarrhea after five days or oral Clindamycin. Thus she was changed to oral doxycycline BID for three weeks. Her infection was found to travel through the flexor hallucis longus tendon sheath. Her course of care was uneventful and this lesion was completely healed in 49 days.
After she realized the need to keep dressings intact and appropriate shoe gear in place, her toe healed uneventfully. Total duration from the inception of her case was approximately 90 days. Her vascular status was surprisingly quite good with an ABI of 0.96 and toe pressures of 110mm Hg.
This patient had ABI of 0.31 with a toe pressure of 40mm Hg and no known run off on his angiogram. His tissue biopsy culture report revealed MRSA and S. Epidermidis, both resistant to Clindamycin and Gentamycin. The organisms were isolated as sensitive only to Vancomycin and sulfamethoxazole and trimethoprim. Although slow to heal, his infected soft tissue lesions did resolve; the first one in 97 days, the second one in 111 days and the third one in 140 days. No amputation was performed.
The location became infected and she failed to keep follow-up appointments for 10 days. Her daughter found her at home with a raging cellulitis and her HMO refused her admission to hospital. Her distal segmental by-pass did clot off during her cellulitic episode. The hardware and fractured bone was removed surgically on an outpatient basis. She achieved complete closure without amputation in 162 days. Of significance, her culture report showed no oral agent which would treat her MRSA or enterococcus.
The literature shows good evidence of why triple helix arranged type I collagen can act as a sustained release vehicle and that many drugs can apparently be delivered on a linear zero order release kinetic basis.
It was not possible, in this study, to measure tissue antibiotic levels. However, previous research [11] has shown how there is minimal, if any, of the toxic drug absorbed into the blood stream. This is despite evidence of achieving tissue levels that cannot be achieved through systemic administration of potent drugs like gentamycin (due to the ototoxicity and nephrotoxicity associated with this drug).
This article illustrates the treatment and care of significant infections involving eight different organisms and three different drugs. The numbers presented here are too small for any scientific conclusions to be drawn from this.
The author is well aware that further laboratory and clinical work is needed to provide the scientific evidence required to create specific outcome results associated with different infections like those presented here. As a rural practitioner, this limits the author from obtaining the large population base needed to really expand this out further.
Thanks to Bill Danzeisen DPM, for scanning the photographs.